What Does Body Age Mean and Can You Actually Lower It?
You’ve probably come across it somewhere: a gym scale that spits out a “metabolic age,” a wellness quiz that tells you your body is 43 when your actual age is 37, or an app that promises to calculate your “real age” from a few health inputs. Most people notice the number, feel vaguely curious or vaguely alarmed, and move on without really understanding what it represents.
Body age — also called biological age — is a genuinely useful concept. The problem is that most of the tools that introduce people to it are measuring something much simpler than what the science actually describes.
The real version is worth understanding, because it’s considerably more specific — and more useful — than what those quizzes deliver.
The Short Summary
- Body age reflects how fast your cells are actually aging, not how long you’ve been alive.
- Most consumer body age calculators compare your health metrics to population averages — they’re not measuring cellular aging directly.
- The biomarkers most closely linked to true biological aging include DNA methylation patterns, telomere length, chronic inflammatory load, and intracellular NAD+ levels.
- Body age can diverge from chronological age in both directions, and several of the markers driving that gap are directly measurable and addressable.
CLIA-Certified Blood Panel
48 biomarkers that reveal your cellular age from home.

Body Age Is Not What Most Calculators Are Measuring
There’s a real concept hiding inside most body age tools. But what those tools actually calculate is a different, much blunter thing.
Chronological Age vs. Body Age
Chronological age is the number of years you’ve been alive. It’s also the number that shows up as a primary risk factor in most clinical guidelines for chronic disease. That correlation is real — the longer you’ve been alive, the more accumulated cellular damage you’re likely to carry.
But it’s an average, not a certainty. Two people born the same year can be in meaningfully different cellular condition. Same genetic background, similar lifestyle choices, and one of them is functioning five years ahead of their age on the inside. The other is behind. Chronological age can’t tell you which one you are.
Body age — also called biological or physiological age — reflects the actual condition of your cells and tissues. It accounts for accumulated cellular damage, DNA expression patterns, genetic factors, how well your metabolic systems are running, and a range of other things a birthday can’t capture. It’s closer to what’s actually happening in your body right now.
What Most Body Age Tools Actually Do
The majority of body age calculators — from gym scales to wellness apps to some online “biological age” tests — work by comparing your health metrics against population averages for your age group. If your blood pressure, cholesterol, BMI, or resting heart rate sit above the average for your demographic, your body age score comes back older. Below average, and it comes back younger.
This isn’t useless. Risk factor awareness matters. But comparing your numbers to a population average is not the same thing as measuring how fast your cells are actually aging. Two people with identical risk factor scores can have meaningfully different biological ages at the cellular level. That’s why researchers have spent decades developing more specific measurement tools.
If persistent fatigue or low energy has you questioning whether your standard labs are giving you the full picture, there are specific biomarkers that standard blood tests routinely miss.
What’s Actually Happening Inside Your Cells
Real biological aging shows up at the molecular level: chemical marks accumulating on DNA, protective caps wearing down at the ends of chromosomes, inflammatory signals building in tissue, cellular energy systems losing efficiency. These are the mechanisms behind the aging process, and they’re what precision measurement actually tracks.
DNA Methylation and the Epigenetic Clock
Every time your cells divide, small chemical compounds called methyl groups attach to specific sites on your DNA. This process — DNA methylation — doesn’t alter your genetic sequence. What it does is change how genes are expressed, turning some on and some off. Over time, those methylation patterns shift in ways that correlate strongly with aging.
Researchers have used this to build what are called epigenetic clocks: models that estimate biological age from methylation data collected at hundreds or thousands of genomic sites. The best-known include the Horvath clock, PhenoAge, and GrimAge.
A 2022 review in Ageing Research Reviews concluded that these clocks correlate with morbidity, mortality, and age-related disease risk, and that they respond measurably to lifestyle interventions.[1]
Epigenetic clocks are currently the most validated single-marker approach to estimating biological age.
Telomere Length and Cellular Wear
Telomeres are the protective sequences at the ends of chromosomes — think of the plastic tips on shoelaces. Each time a cell divides, those tips get slightly shorter. When they get too short, the cell either stops dividing or initiates a stress response.
Shorter telomeres track with higher rates of age-related disease and earlier mortality. A 2021 review in Frontiers in Genetics found that telomere length is most informative when used alongside other biomarkers rather than as a standalone measure — as a single data point, it gives only a rough estimate of aging rate.[2]
No single marker tells the whole story. Multi-marker approaches consistently outperform single tests in research on biological age, and this is a good example of why.
Inflammation and Cellular Senescence
As cells age, some stop dividing but don’t die. These senescent cells, sometimes called “zombie cells,” don’t just go quiet. They secrete a mix of inflammatory proteins called the senescence-associated secretory phenotype (SASP) that can push neighboring cells toward the same state. The result is a kind of chronic, low-grade systemic inflammation researchers call “inflammaging.”
A 2023 review in Signal Transduction and Targeted Therapy describes inflammaging as one of the core drivers of biological aging, with direct connections to neurodegeneration, cardiovascular disease, and metabolic decline. The accumulation of senescent cells and the inflammatory load they produce is one of the clearest signs that body age is pulling ahead of chronological age.[3]
NAD+ Decline and Cellular Energy
NAD+ (nicotinamide adenine dinucleotide) is a molecule your cells rely on to produce energy, repair DNA, and regulate hundreds of metabolic processes. It declines steadily with age, with measurable drops often starting in the 30s.
That decline matters because NAD+ is involved in several biological aging pathways simultaneously. Lower NAD+ means less capacity for mitochondrial energy production, slower DNA repair, and upregulated inflammatory signaling.
A systematic review covering 147 studies in Experimental Gerontology found that declining NAD+ levels track closely with age-related degenerative processes, and that restoring those levels produces measurable effects in both preclinical and early clinical research.[4]
What makes NAD+ worth particular attention here is that it’s directly measurable. Unlike some aging biomarkers that require tissue biopsy or complex genomic analysis, intracellular NAD+ can be assessed from a blood sample — a clear readout of where that specific pathway stands.
If you want context on how mitochondrial supplements factor into cellular energy production, that’s a useful place to start.
How Body Age Is Actually Measured
The gap between a gym scale readout and a clinical biological age assessment is significant. Here’s what more precise measurement actually involves.
Epigenetic Clock Testing
Dedicated epigenetic age tests analyze DNA methylation patterns from a blood or saliva sample, then run the data through an established clock algorithm to generate a biological age estimate. These tests are more expensive than standard bloodwork, and predictive accuracy varies by which model is used.
A 2025 review in Frontiers in Cardiovascular Medicine notes that advances in omics technologies and biomarker research are improving the precision of biological aging assessments, with integration of multiple data streams producing more accurate and useful results than any single marker alone.[5]
Blood Biomarker Panels
Comprehensive blood panels that include inflammatory cytokines, senescence markers, NAD+ levels, longevity proteins like Klotho, and metabolic indicators give a multi-system picture of where biological aging actually stands. The value comes from looking across pathways — not just one.
A panel that measures only one or two markers will miss most of what’s driving body age. The hallmarks of aging interact with each other. Mitochondrial dysfunction drives NAD+ depletion. Cellular senescence amplifies inflammatory load. Inflammaging suppresses NAD+ metabolism. A single data point doesn’t give you an accurate picture.
What Makes a Biomarker Worth Measuring
Researchers use fairly specific criteria to evaluate aging biomarkers: they should predict health outcomes better than chronological age alone, reflect the actual mechanisms of aging rather than secondary effects of disease, be measurable repeatedly without harm, and respond to interventions in a trackable way.
Biomarkers that meet these criteria — including epigenetic methylation patterns and intracellular NAD+ — are the ones that generate useful information.
Understanding how to test NAD+ levels is a reasonable first step for anyone starting to explore their own cellular picture.
What Affects Your Body Age — and How to Improve It
The factors that drive body age aren’t abstract — they operate directly on the cellular mechanisms described above. Here’s what the research actually supports, and how to improve your body age across each lever.
How to Improve Your Body Age
Exercise
- Regular physical activity supports telomere maintenance and lowers systemic inflammatory markers.
- Resistance training improves body composition, helps preserve muscle mass, and supports NAD+ metabolic efficiency.
- Regular exercise at moderate intensity — around 150 minutes per week — shows measurable effects on epigenetic aging patterns in clinical studies.
Sleep quality
- Chronic sleep disruption is one of the fastest accelerants of epigenetic aging in the literature.
- Poor sleep elevates inflammatory cytokines, which compounds senescent cell accumulation over time.
- Consistent, restorative sleep is one of the few lifestyle choices that moves multiple aging pathways at once.
Diet and metabolic health
- A balanced diet low in processed foods and refined sugars and high in vitamins and minerals reduces body fat and lowers chronic inflammatory load directly.
- Caloric restriction and intermittent fasting show effects on NAD+ metabolism and cellular autophagy — the process by which cells clear out damaged components.
- Blood sugar dysregulation accelerates glycation damage independently of other risk factors, adding measurable years to biological age on its own.
Stress management
- Prolonged psychological stress correlates with accelerated telomere shortening and elevated senescence markers.
- Chronic cortisol elevation suppresses NAD+-dependent repair mechanisms over time.
- Exercise, sleep, and mindfulness all show overlapping effects on inflammatory and epigenetic aging markers — managing stress and managing body age are the same project.
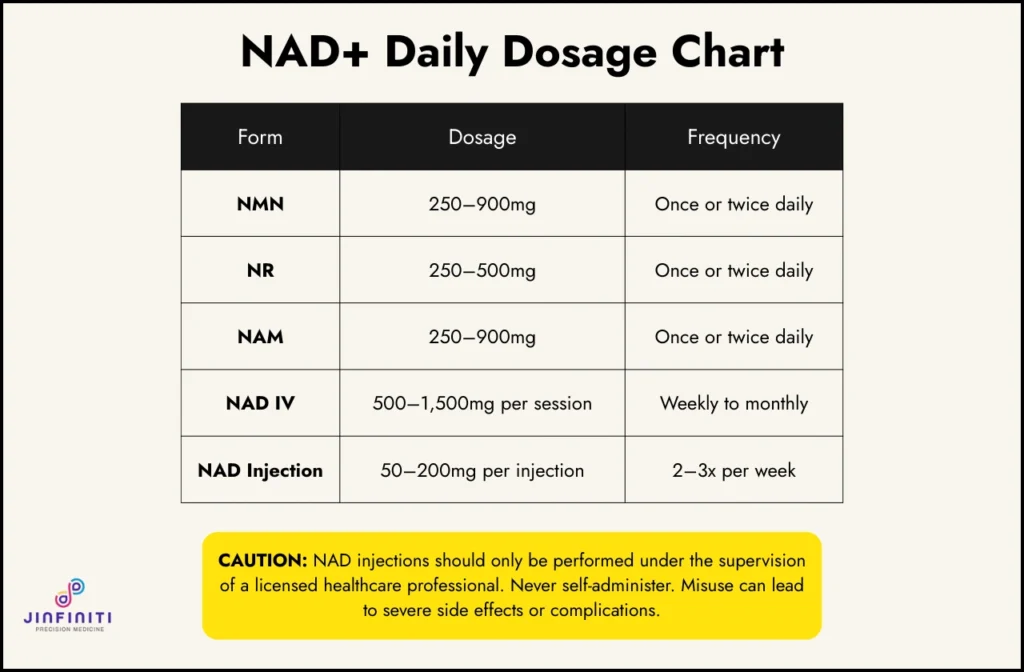
NAD+ support
- NAD+ can be measured directly, which means it’s one area where you can verify whether an intervention is actually working rather than assuming it is.
- Unlike most lifestyle interventions, targeted NAD+ precursor supplementation allows you to track a specific molecular readout before and after — not just how you feel.
Why Measuring Before Acting Matters
Most people try things and hope for improvement. That’s reasonable as far as it goes — but it doesn’t tell you which pathways are actually depleted in your particular biology.
Someone with measurably low NAD+ and normal inflammatory markers has a different cellular picture from someone with elevated senescence markers and adequate NAD+. The right approach for each is different. Without a baseline, you’re applying a generic strategy to a specific problem — and you won’t know whether it worked.
🧬 RELATED READING
- Body age pulling ahead of chronological age has specific causes — and early warning signs worth knowing. Read about what drives premature aging.
- NAD+ is one of the most studied cellular aging markers. Here’s what the evidence actually shows about NMN benefits for cellular health.
- If you’re considering NAD+ support, understanding how to approach NAD+ dosage and timing is a useful first step.
Getting a Clearer Picture of Your Body Age
The question people are really asking when they encounter body age calculators is: is my body holding up the way it should be? The frustration isn’t with the question. It’s that most available tools aren’t built to actually answer it.
A longevity panel like AgingSOS that includes NAD+, senescence markers, inflammatory cytokines, Klotho, and metabolic indicators doesn’t just return a number. It shows which specific systems need attention, which are functioning within range, and where to start.
“Most people come to us after they’ve already tried the obvious answers,” says Dr. Jin-Xiong She, founder of Jinfiniti Precision Medicine. “What body age really means at the cellular level is whether your systems are being maintained, or are slowly losing ground. Testing gives you that answer. Without it, you’re managing your health without knowing which problem you’re actually solving.”
Body age as a concept only becomes useful when you can measure the things behind it. A quiz score doesn’t change anything. Knowing your NAD+ is suboptimal, your inflammatory markers are elevated, or your Klotho is low — that gives you somewhere to start.
Frequently Asked Questions
What Is the Difference Between Body Age and Biological Age?
The terms are used interchangeably in most contexts. Body age and biological age both refer to how your cells and tissues are functioning compared to chronological age norms. “Body age” tends to appear in consumer-facing tools (gym scales, apps), while “biological age” is more common in clinical and research settings. Same concept, different context.
Can Body Age Be Lower Than Your Chronological Age?
Yes, and it happens more than people expect. People with consistently healthy habits, well-maintained NAD+ levels, low chronic stress, and good sleep regularly show biological ages below their chronological age on panel testing. The relationship between how long you’ve lived and how fast you’ve aged is not fixed.
What Is a Normal Body Age?
There’s no single universal standard. Biological age assessments compare your markers to reference ranges built from population data. The specific number matters less than whether your key markers are trending in the right direction — and whether the interventions you’re making are actually moving them.
Can You Reverse Your Body Age?
The evidence supports slowing the rate of biological aging and, in some cases, improving specific markers meaningfully. “Reversing” is probably stronger language than most clinical research warrants. Epigenetic clocks have shown responsiveness to lifestyle and supplementation interventions in controlled studies.
What Is the Most Accurate Way to Measure Body Age?
Epigenetic clock testing is currently the most validated single approach. In practice, multi-marker blood panels that include NAD+, inflammatory cytokines, senescence markers, and longevity proteins like Klotho give a more complete and useful picture than any single test. The goal isn’t a single number — it’s understanding which systems are on track and which need attention.