What Is NAD and Why Does It Matter for Your Energy?
What You Should Know
- NAD is a coenzyme that turns food into cellular energy and supports DNA repair
- Your levels naturally decline with age—often beginning in your 30s
- Low NAD contributes to fatigue, brain fog, and slower recovery
- Without testing, you can’t know if your levels are deficient or if supplementation is working
When exhaustion doesn’t respond to the standard fixes, it often points to something happening at the cellular level. One of the most common and underrecognized drivers is a decline in NAD, a molecule your cells rely on to produce energy.
NAD — short for nicotinamide adenine dinucleotide — exists in every living cell in your body. It’s not a vitamin or a hormone. It’s a coenzyme, which means it helps other molecules do their jobs. Without sufficient NAD, your cells can’t convert the food you eat into usable energy. They also can’t repair DNA damage, respond to stress, or regulate metabolism effectively.
The problem is that NAD levels decline measurably with age, often beginning in your 30s or 40s. Research shows that many people experience a 40-50% drop by age 50. And unlike most biomarkers, NAD can be measured directly — which means you can know exactly where your levels stand and take action based on data, not guesswork.[1]
What NAD Actually Is
NAD is a small molecule found in every living cell, from bacteria to humans. It’s been studied for over a century — first discovered in 1906 during fermentation research — but our understanding of its importance in human health has deepened significantly in recent decades.
At a chemical level, NAD is made of two nucleotides joined by phosphate groups. One contains adenine, the other nicotinamide. The body can produce NAD from vitamin B3 (niacin) or the amino acid tryptophan, though most NAD is recycled through a salvage pathway rather than built from scratch.
What makes NAD essential is its ability to shuttle electrons between molecules. This electron transfer is the basis for nearly all energy production in your body.
The Two Forms: NAD+ and NADH
NAD exists in two forms that constantly cycle back and forth: NAD+ and NADH.
NAD+ is the oxidized form — think of it as an empty shuttle bus, ready to pick up electrons during metabolic reactions. NADH is the reduced form — the shuttle carrying electrons that can be used to generate ATP, your cells’ energy currency.
Together, these forms create a loop. NAD+ accepts electrons from nutrients like glucose and fat, becoming NADH. NADH then delivers those electrons to the mitochondria, where they drive ATP production. Once the electrons are dropped off, NADH converts back to NAD+, and the cycle continues.
This constant cycling is what keeps energy flowing in your cells. When NAD levels are too low, the loop slows down. Cells struggle to produce enough ATP, and you feel it as fatigue, slower recovery, or cognitive fog.
Why NAD Matters: What It Does in Your Body
NAD is involved in hundreds of cellular processes. Studies show it participates in more than 500 enzymatic reactions.[2]
A few roles stand out as particularly critical:
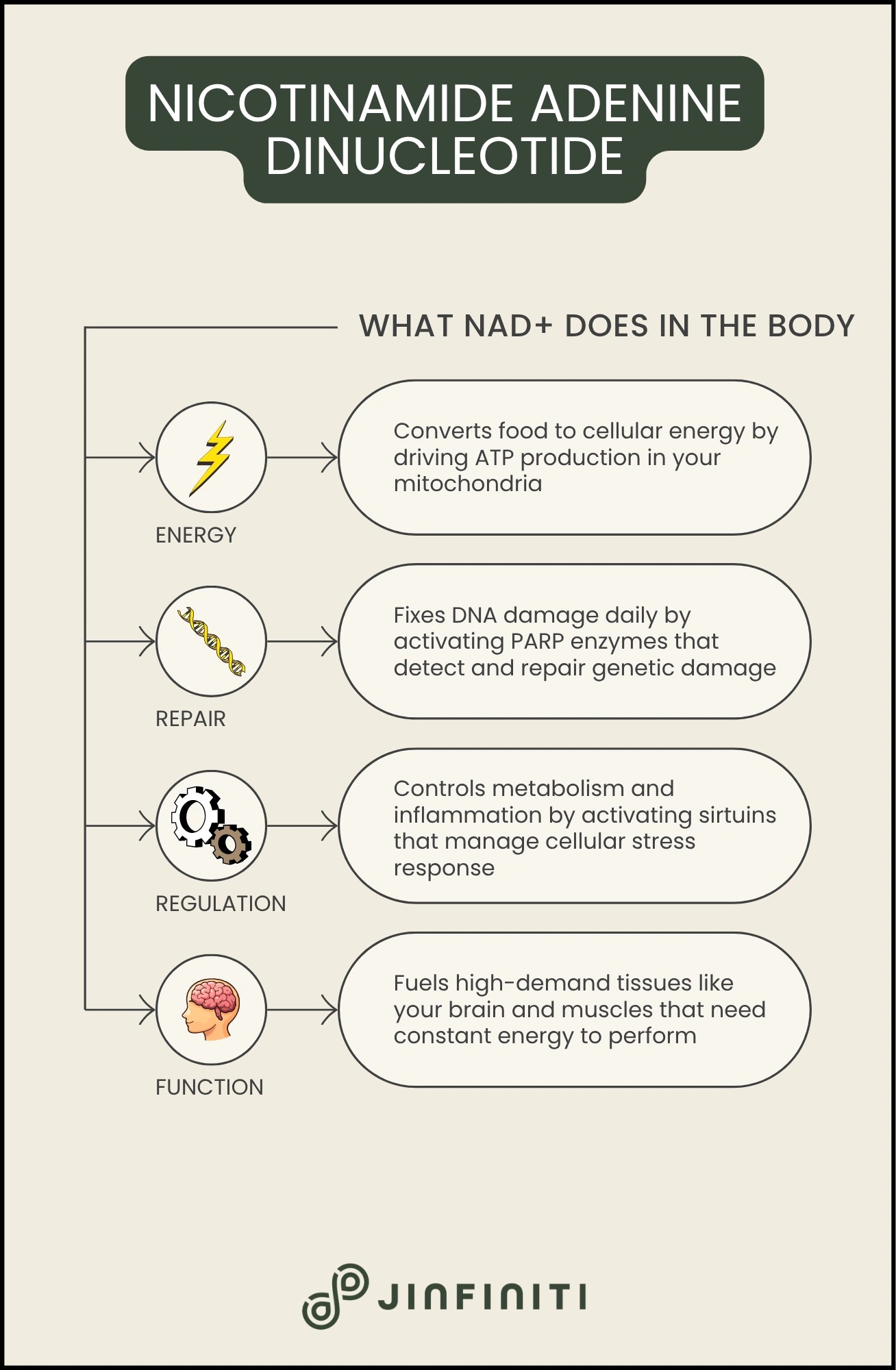
Energy production: Inside your mitochondria, NAD+ drives the reactions that convert glucose and fatty acids into ATP. Without sufficient NAD, mitochondrial function declines, and cellular energy production becomes less efficient.
DNA repair: Every day, your cells accumulate DNA damage from normal metabolism, UV exposure, and environmental toxins. NAD is consumed by enzymes called PARPs that detect and repair this damage. When NAD levels are low, DNA repair slows, and damage accumulates faster.
Metabolic regulation: A family of proteins called sirtuins only function in the presence of NAD. Sirtuins influence everything from fat storage to inflammation to circadian rhythm. They’re often called “longevity genes” because of their connection to healthy aging and stress resistance.
Brain function: Your brain represents only about 2% of your body weight but consumes roughly 20% of your total energy. NAD supports the high metabolic demands of neurons, and declining NAD in the brain has been linked to cognitive decline and neurodegenerative conditions.[3]
This isn’t just about “anti-aging.” NAD is how your body functions today. When levels are optimal, cells repair themselves efficiently, produce energy consistently, and respond to stress effectively. When levels drop, those processes slow — and you feel the difference before any disease state appears.
Why NAD Levels Decline With Age
NAD decline is one of the most consistent findings in aging research. It’s been documented in rodents, primates, and humans across multiple tissues.
Human studies show that whole blood NAD levels decline significantly with age, particularly in men. Women’s levels tend to fluctuate more after age 50, likely due to hormonal changes. In skeletal muscle, the decline can be anywhere from 15% to 65%, depending on the individual and the measurement method. Brain NAD also decreases measurably, which correlates with reduced cognitive function.[1]
The decline happens through two main mechanisms.
Reduced production occurs because the enzymes responsible for making NAD become less efficient with age. The key enzyme, NAMPT, decreases in activity over time. This creates a bottleneck — your cells can’t produce NAD as quickly as they once did, even if you’re consuming adequate precursors through diet.[4]
Increased consumption happens because certain NAD-degrading enzymes become more active as you age. One called CD38 rises in response to chronic inflammation. Another group, PARPs, ramps up activity in response to accumulated DNA damage. Both consume NAD to do their jobs, which depletes the cellular pool.[5][6]
Think of it as a sink where the faucet slows while the drain widens. Less NAD coming in, more NAD going out. Over time, this leads to the net decline that begins earlier than most people realize.
What Drives NAD Decline?
Age is the primary driver, but several factors accelerate the process:
- Chronic inflammation increases the activity of CD38 and other NAD-consuming enzymes[5]
- Metabolic stress from a high-fat or high-sugar diet can reduce skeletal muscle NAD levels in just two months[7]
- Obesity is associated with both lower NAMPT expression and higher PARP activity, creating a compounding effect[8]
- DNA damage from UV exposure, toxins, or normal cellular metabolism forces PARPs to consume more NAD for repair[6]
If you’re dealing with persistent fatigue despite normal blood tests, NAD deficiency is one of the cellular-level explanations that standard panels miss.
Signs Your NAD Levels May Be Low
NAD decline doesn’t announce itself with a single, obvious symptom. Instead, it shows up as a pattern of issues that conventional testing often fails to explain.
Common signs of low NAD include:
- Persistent fatigue that doesn’t resolve after adequate sleep — not the kind that goes away after a good night’s rest, but the kind that lingers despite doing everything “right”
- Slower recovery from exercise, illness, or stress — when NAD levels are optimal, cells repair damage efficiently and bounce back quickly; when levels are low, recovery takes longer
- Brain fog and difficulty concentrating — neurons are metabolically demanding, and when NAD drops, cognitive performance often follows
- Metabolic changes — difficulty maintaining weight, worsening insulin sensitivity, or changes in how your body responds to food
If you’re still tired after 8 hours of sleep, cellular energy deficiency is worth investigating.
The challenge is that these symptoms overlap with many other conditions. Thyroid disorders, sleep apnea, anemia, hormonal imbalances, and dozens of other issues can present similarly. That’s why symptom-based guessing rarely leads to a clear answer.
Measurement is the only way to know.
The NAD Measurement Problem
Most people who take NAD supplements have no idea if they’re actually working.
The wellness industry has embraced NAD precursors like NMN and NR over the past few years. Hundreds of products now claim to boost NAD levels, often with impressive-sounding promises about energy, longevity, and cognitive performance. But very few people taking these supplements ever test their levels — before, during, or after.
That’s a problem, because NAD metabolism is highly individual. The same dose that optimizes one person’s levels might do almost nothing for another. Some people absorb precursors efficiently; others don’t. Some have high baseline consumption from inflammation or DNA damage; others don’t. Without testing, you’re guessing.
Intracellular measurement matters because that’s where NAD actually works. Some tests measure NAD in blood plasma, which can give you a general sense of systemic levels. But the NAD inside your cells — where energy production, DNA repair, and metabolic regulation happen — is what drives outcomes. Intracellular NAD testing provides a much more accurate picture of your cellular energy status.
Dr. Jin-Xiong She’s clinical research established clear benchmarks for interpreting results. Optimal NAD levels fall between 40 and 100 micromolar (μM). Anything below 40μM is considered suboptimal or deficient. Levels above 100μM may not provide additional benefits and could potentially cause issues.
These ranges give you a concrete target, not a vague reference to “normal.”
Why Testing Changes Everything
Jinfiniti developed the world’s first consumer-grade intracellular NAD test in 2019. Before that, NAD testing was largely confined to research labs. Now, you can measure your baseline, track changes over time, and personalize your approach based on data.
Here’s what measurement enables:
- Establishing your baseline tells you where you’re starting from — you might discover your levels are already optimal, in which case supplementation isn’t needed, or you might find they’re severely deficient, which explains symptoms that have gone undiagnosed for years
- Validating whether supplementation works removes the guesswork — some people respond well to NMN, others respond better to niacinamide or NR, and testing shows you what’s actually happening in your cells, not what a bottle label promises
- Personalizing your dosing is critical because there’s no one-size-fits-all approach to NAD optimization — in Jinfiniti’s clinical trial, 85% of participants reached optimal NAD levels within four weeks, but that success rate depended on personalized dosing based on measured results, not generic recommendations
- Tracking progress over time lets you adjust as needed — NAD levels aren’t static; they respond to changes in diet, exercise, stress, inflammation, and supplementation, so retesting every few months ensures you’re maintaining optimal levels rather than drifting back into deficiency
As Dr. She puts it: “Measure before acting. NAD optimization isn’t about taking the same dose as someone else and hoping it works. It’s about knowing your levels, acting on that information, and confirming the results. Precision requires data.”
Without measurement, you’re flying blind. With it, you can make informed decisions and track real progress.
How to Support Healthy NAD Levels
If you’re looking to optimize your NAD levels, the most effective approach combines testing with targeted intervention.
Here’s what works:
- Test first — Establish your baseline with an intracellular NAD test; this removes the guesswork and tells you whether your levels are deficient, suboptimal, or already optimal
- NAD+ precursor supplementation is the most direct way to raise levels — NMN and NR are the most studied precursors; both convert to NAD inside your cells, though they follow slightly different pathways; niacinamide (a form of vitamin B3) also works through the salvage pathway
- Lifestyle factors can support NAD production — regular exercise, moderate caloric intake, and consistent sleep all influence NAD metabolism, though they’re unlikely to fully counter age-related decline on their own
- Reduce chronic inflammation through diet, stress management, and addressing underlying health issues — this helps by lowering NAD consumption from enzymes like CD38
- Retest and adjust after 4-6 weeks — if your levels haven’t reached the optimal range, adjust your dose or try a different precursor; if they have, continue monitoring periodically to ensure they stay stable
Jinfiniti’s Vitality NAD+ Booster uses a multi-pathway formula that combines NMN, niacinamide, creatine, and D-ribose. The clinical trial data showed an average doubling of NAD levels within four weeks, with 85% of participants reaching the optimal range.
The goal isn’t to chase an arbitrary number. It’s to restore your cells’ ability to produce energy efficiently, repair damage effectively, and respond to stress appropriately. When NAD levels are optimal, you feel it — and the data confirms it.
The Bottom Line
NAD is not a wellness trend. It’s a fundamental molecule that’s been studied for over a century and plays a documented role in energy production, DNA repair, and metabolic regulation. Levels decline measurably with age, and that decline has real consequences — fatigue, slower recovery, cognitive fog, and increased vulnerability to age-related disease.
The difference between vague health advice and precision medicine comes down to measurement. You can’t optimize what you don’t measure. And without data, you’re guessing whether your NAD levels are deficient, whether supplementation is working, or whether you even need it in the first place.
If you’re dealing with unexplained fatigue, cognitive decline, or signs of accelerated biological aging, NAD testing gives you a clear starting point. It’s not the only factor that matters, but it’s one you can measure, address, and track over time.
Frequently Asked Questions
Is NAD the same as vitamin B3?
NAD is a molecule your body makes from vitamin B3 (which includes niacin, niacinamide, and nicotinamide riboside). Vitamin B3 is a precursor — a building block your cells use to produce NAD. You can get vitamin B3 from food or supplements, but your body still needs to convert it into NAD through metabolic pathways. Learn more about the differences between NAD, niacin, and niacinamide.
Which foods contain NAD?
Very few foods contain NAD directly. Instead, you get NAD precursors from foods rich in vitamin B3 — like meat, fish, mushrooms, peanuts, and fortified grains — or from foods containing tryptophan, an amino acid your body can convert to NAD. However, dietary sources alone rarely raise NAD levels significantly, especially after age 40. See the full list of NAD-boosting foods.
Are NAD supplements safe?
NAD precursors like NMN, NR, and niacinamide are generally well-tolerated in clinical studies, with mild side effects like nausea or flushing reported in some people. Most research shows a favorable safety profile for short-term use. However, long-term safety data is still limited, and it’s best to work with a healthcare provider — especially if you have underlying health conditions.
Can NAD help with weight loss?
NAD doesn’t directly cause weight loss, but it plays a role in metabolic function and energy regulation. Some research suggests NAD precursors may support metabolic health, particularly when combined with exercise and caloric moderation. However, NAD supplementation is not a weight-loss drug, and results vary widely between individuals. Read more about NAD, NMN, and weight loss.
What is the difference between NAD+ and NADH?
NAD+ is the oxidized form of the molecule, ready to accept electrons during metabolic reactions. NADH is the reduced form, carrying electrons that can be used to generate ATP. They cycle back and forth continuously, and both are necessary for cellular energy production.
Can you increase NAD levels naturally without supplements?
Lifestyle factors like exercise, caloric moderation, and adequate sleep can support NAD production to some degree. However, research suggests that lifestyle changes alone are often insufficient to fully counter the age-related decline in NAD levels, particularly after age 40. Precursor supplementation is typically needed for meaningful increases.[2]
What are optimal NAD levels?
Based on Dr. Jin-Xiong She’s clinical research, optimal intracellular NAD levels fall between 40 and 100 micromolar (μM). Levels below 40μM are considered suboptimal or deficient. Levels above 100μM may not provide additional benefits and could potentially be harmful.
How do I know if my NAD levels are low?
The only reliable way to know is through testing. Symptoms like persistent fatigue, brain fog, and slow recovery can suggest low NAD, but they overlap with many other conditions. Intracellular NAD testing provides a precise measurement of your cellular levels.
Does NAD supplementation work for everyone?
NAD precursors can raise levels in most people, but individual responses vary widely. Some people absorb and convert precursors efficiently, while others need higher doses or different forms. Without testing, you won’t know if a supplement is actually working for you. That’s why personalized dosing based on measured levels is more effective than generic recommendations.
How long does it take to see results from NAD supplementation?
Most people who respond to NAD precursors notice changes within 2-4 weeks. Clinical studies, including Jinfiniti’s trial with Vitality NAD+ Booster, show measurable increases in NAD levels within four weeks. However, subjective improvements in energy, recovery, and cognitive clarity may appear sooner or later depending on your baseline levels and overall health.
Does NAD interact with GLP-1 medications?
NAD and GLP-1 pathways both influence metabolic health, and there’s emerging research on how they interact. If you’re using GLP-1 medications, NAD optimization may offer complementary benefits. Learn more about the relationship between NAD and GLP-1.